Choksi Laboratories Receives Enhanced CDSCO Approval for Medical Devices Quality Assessment
We are thrilled to announce that Choksi Laboratories Limited (CLL) has received enhanced scope approval from the Central Drugs Standard Control Organization (CDSCO) for the testing and evaluation of medical devices. This expansion strengthens our commitment to ensuring high-quality and regulatory-compliant medical devices for the healthcare industry.
Expanded Scope of Testing
The approval now covers 59 medical devices across various classes, allowing CLL to conduct comprehensive chemical, microbiological, and elemental analysis. Some of the critical quality parameters included in our expanded scope are:
- Chemical Characterization – Identification of the chemical composition of medical devices.
- Extractables and Leachables Assessment – Evaluating potential chemical compounds that may migrate from the device to the patient.
- Sterilization Residues Testing – Detection of Ethylene Glycol (EG), Diethylene Glycol (DEG), Ethylene Oxide (EO), and Ethylene Chlorohydrin (ECH) to ensure product safety.
- Elemental Analysis – Determining the presence of extractable and total elements that could impact device performance and safety.
CLL already had an approved scope for microbiological testing of medical devices. With this enhanced scope, we are now positioned to provide a comprehensive suite of quality assessment services for a broader range of medical devices.
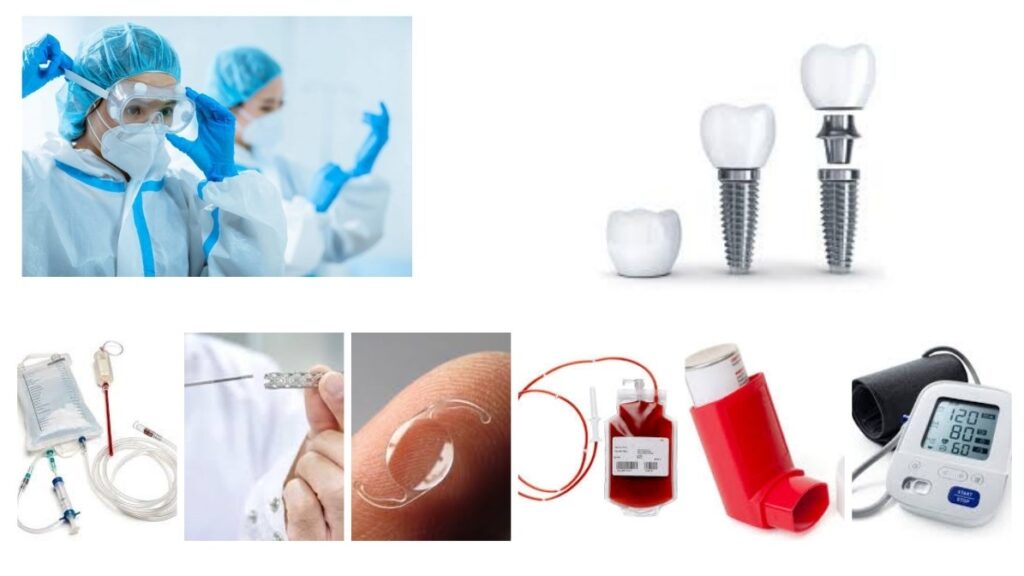
Medical Devices Covered in the Expanded Scope
Our state-of-the-art laboratory can now test and evaluate a diverse range of medical devices, including but not limited to:
- IV Bags and Sets
- Catheters
- Lenses & Contact Lenses
- Dental Implants and Orthopedic Implants
- Cardiovascular Devices and Stents
- Surgical & Non-Surgical Gloves
- Intrauterine Devices (IUDs)
- Nebulizers, Aspirators, and Inhalers
- Face Masks, Nasal Masks, and Oral Masks
Commitment to Quality and Compliance
At Choksi Laboratories, we uphold the highest standards of testing and regulatory compliance. Our advanced analytical capabilities, combined with our expertise in pharmaceutical and medical device testing, ensure that we deliver reliable, accurate, and regulatory-compliant results.
With this CDSCO-approved enhanced scope, we reaffirm our commitment to supporting medical device manufacturers, importers, and healthcare providers in delivering safe and effective medical solutions to the market.
🔬 For inquiries about our medical device testing services, contact us today!